Parasite transmission is dependent upon host diet and habitat use. A recent paper in the Journal of Animal Ecology examined over 350 species of lizard in search of a link between parasite diversity and lizard ecology. Here to tell us more is lead author Dr Tommy Leung, a Lecturer in Parasitology and Evolutionary Biology at the University of New England, Australia.
All animals are infected with parasites of some kind, in fact at any one time an animal might be infected with multiple species of different parasites. But some animals have more parasites than others, so why is that? To find out, in this study, Dr Janet Koprivnikar and I decided to compare the communities of parasitic worms living in lizards to try and figure out why some lizards are more wormy than others.
We wanted to see if the number of parasitic worms that infect a given lizard species is somehow associated with aspects of their ecology and life history, including their diet, habitat, body size, whether they lay eggs or give birth to live young. To do that, we collected data on published parasite records of over 350 species of lizards, along with information on their ecology and life history.

Why lizards? Well, lizards are very diverse – there are over 6000 known living species and they represent 60% of all known living reptiles. Most existing studies which looked for relationships between host characteristics and parasite communities have focused on birds, mammals, and fish – because they happen to be the animal groups which have the most well-documented parasite records. While such studies have provided us with some interesting insight into the factors that influence parasite diversity and abundance, by focusing on just those three host groups, they have neglected a big part of living vertebrate diversity and provide an incomplete picture of factors that determine patterns of parasitic infections.
So what did find? Well, out of all the factors we looked at, diet was by far the most influential. We found that herbivorous lizards have fewer parasite species than the omnivorous and carnivorous ones. A possible reason for that is many parasitic worms have complex life cycles and use small animals as vehicles to reach their host. So the more animals a lizard consumes, the more likely it would end up swallowing some parasites. Conversely those that don’t feed on animals wouldn’t encounter those parasites.
In addition to being the final host for the adult stage of a range of parasitic worms, lizards are also host to some larval parasites which in turn use them as vehicles to reach their final hosts – these include predatory birds, mammals, or reptiles which would eat lizards. So lizards play different roles in the life cycles of many different parasites.
One unexpected outcome of our analysis was that we found lizards with arboreal or climbing habits tend to harbour more larval parasites. So why would the propensity to climb, of all things, make a lizard more susceptible to hosting parasite larvae? Well, maybe it’s not their habitat as such which is causing this, but something else to do with the ecology of those lizards. When we looked at the diet of those arboreal species, 86% of them were carnivorous, and none were herbivorous. So it’s not the climbing itself that does it, but what those climbing lizards eat which might be causing this pattern.
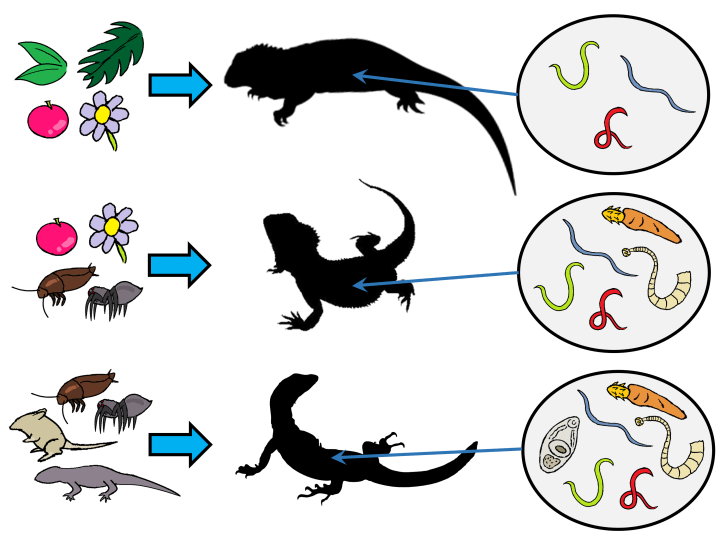
Something else which emerged from our analysis is that lizard species that use aquatic habitats tend to harbour more parasitic flukes. Flukes (digenean trematodes) have complex life cycles which involves many different host animals, but they always have a stage where they undergo asexual proliferation (essentially cloning themselves) and that usually takes place in an aquatic snail. Through this process, they produce free-swimming larval stage which then infect the next host in the life cycle. This entire part of the life cycle – infecting a snail and releasing free-swimming fluke larvae – require water or at least the presence of moisture. So that might explain why lizards that hang out around water are more likely to be infected with flukes.
However, there might also be a more indirect explanation – lizards do not become infected by flukes through the free-swimming larval stage produced by the snail host, but instead through ingesting animals that are infected by the fluke’s larvae. Prey animals that live in or near water such as aquatic insects, crustaceans, and amphibians often serves as second host for fluke larvae, and it is likely that lizards actually become infected through eating those animals. Once again, it comes down to diet.
What our study demonstrated is that there seems to be a variety of different factors which can influence the parasite communities of lizards, but some of them are linked to each other in some way and cannot be considered to be mutually exclusive.
Aside from host physiology and immunity, an important factor that determines whether a given parasite successfully infects a host is what parasitologist Claude Combe called the “Encounter Filter”. Basically, a host can only get infected by parasites that it comes into contact with, and that depends on whether it lives in areas where the parasite is found and if it eats food items that are likely to contain the parasite’s infective stages.
This also means that animals might readily acquire new parasites if they changes their diet to feed on a new food source. This might be caused by environmental disturbances, such as range expansions driven by climate change, or when new species are introduced to a habitat. With the daily transport of plants and animals around the planet (whether on purpose or accidental), what we are essentially doing is conducting an unintentional global experiment in lifting the “Encounter Filter”, bringing together parasites and hosts that otherwise would have never previously met each other.
In short, you are infected by what you eat, at least when it comes to parasitic worms.
More Info:
Leung and Koprivnikar (2019) Your infections are what you eat: How host ecology shapes the helminth parasite communities of lizards. Journal of Animal Ecology. doi: 10.1111/1365-2656.12934
