This blog post is provided by Jenny Ouyang and tells the #StoryBehindthePaper for the paper “Changes in the rearing environment cause reorganization of molecular networks associated with DNA methylation”, which was recently published in Journal of Animal Ecology. In their paper they explore the impact of urbanisation on DNA methylation in young birds.
Urbanization is one of the most prevailing forms of habitat change, causing biodiversity loss through local extinction processes. Urban expansion is expected to impact a quarter of all endangered species in the next decade. Nevertheless, individuals vary in their response to these drastic changes, with some unable to occupy these new habitats while others persist and thrive.
This difference in the ability to adapt has promoted the study of phenotypic traits that allow individuals to inhabit urban areas. A key trait that can facilitate adaptation is the degree at which animals respond to stressors. This stress response, which includes elevation of baseline circulating concentrations of glucocorticoids, has a heritable component and exhibits individual variation. Epigenetic mechanisms can alter organism function without changes in the DNA sequence, representing a possible mechanism for the observed response to urban stressors.

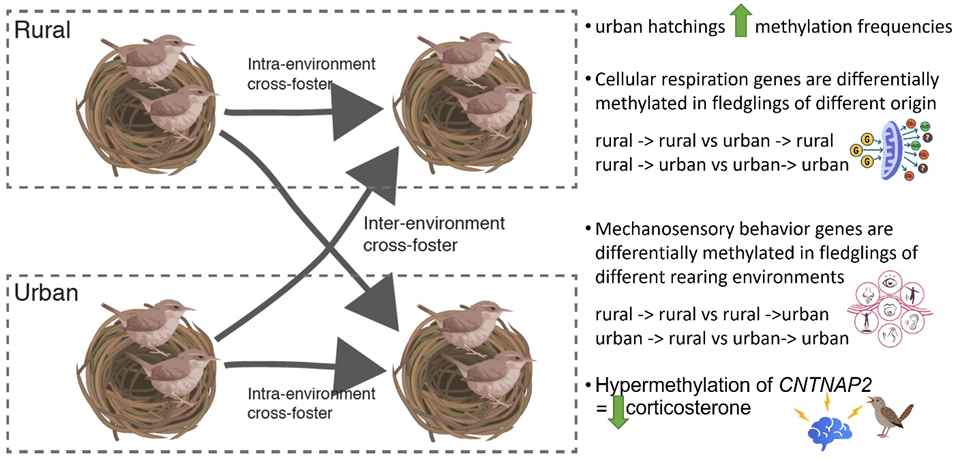
In a recent paper in the Journal of Animal Ecology, we explored the contribution of DNA methylation towards observed urban phenotypes. When house wren parents bred in nestboxes across a network of urban and rural field sites, we moved their offspring across and within sites to disentangle the contribution of genetic and plastic mechanisms to the glucocorticoid phenotype. This type of experiment is an inter- and intra-environmental cross-foster to analyze the contribution of DNA methylation to early-life phenotypic variation.
We observed age-related patterns in offspring methylation, indicating developmental effects of the rearing environment. We further discovered different networks of genes were important at hatching compared to fledging. For example, we found several genes involved in auditory response and learning networks were differentially methylated across experimental treatments. Analyses showed that cellular respiration genes were differentially expressed at hatching and behavioral and metabolism genes were differentially expressed at fledgling. Lastly, hyper-methylation of a single gene (CNTNAP2) is associated with decreased glucocorticoid levels and the rearing environment. Urban house wrens appear to be hypermethylated during hatching compared to their rural counterparts. As offspring aged, wrens that stayed in the same environment increased methylation frequencies but wrens that moved between environments did not show a similar increase. These age and environment-related changes in methylation frequencies suggest that the urban phenotype is a result of both genetic and environmental factors.
Our findings are suggestive that DNA methylation can shape the physiological phenotype and is empirical evidence for a mechanism by which individuals thrive in changing environments. Together, this work provides an unprecedented empirical system that we have leveraged to explore the influence of both genetics and environment on DNA methylation. DNA methylation may be a mechanism by which individuals adjust to novel environments during their lifespan. Understanding the genetic and environmental basis of local adaptation is important in predicting species’ responses to an urbanizing world.
Author bio
Jenny Ouyang – I am an integrative physiologist at the University of Nevada, Reno. I am interested in how animals physiologically adapt to changing environmental conditions.
Read the paper
Read the full paper here: von Holdt, B. M., Kartzinel, R. Y., van Oers, K., Verhoeven, K. J. F., & Ouyang, J. Q. (2023). Changes in the rearing environment cause reorganization of molecular networks associated with DNA methylation. Journal of Animal Ecology, 00, 1– 17. https://doi.org/10.1111/1365-2656.13878