Eco-evolutionary dynamics are well studied but the term is applied to a wide variety of effects and interactions. Yet comparing these different types of studies on eco-evolutionary dynamics will inform on how this field can move forward, which is precisely the aim of a recent British Ecological Society cross-journal Special Feature. Here, Isabel Smallegange (an Associate Professor of Population Biology at the University of Amsterdam) discusses a study published within this Special Feature that investigates how an eco-evolutionary feedback loop between population dynamics and fighter expression affects the evolution of alternative reproductive tactics.
To contribute towards integrating the field of eco-evolutionary dynamics and move the field forward, scientific journals from the British Ecological Society, including the Journal of Animal Ecology, have recently published a cross-journal special feature entitled “The diversity of eco-evolutionary dynamics: comparing the feedbacks between ecology and evolution across scales”, edited by Franziska Brunner, Jacques Deere, Martijn Egas, Christophe Eizaguirre and Joost Raeymaekers *.
How evolutionary changes (like shifts in genotype and phenotype frequencies) and ecological changes (like the size, composition and growth of an animal or plant population) affect each other is a topic of intense and growing investigation in biology. Why? Because for a long time, ecologists ignored evolutionary processes as they were assumed to occur at much longer time scales (thousands to millions of years) compared to ecological processes (days to years). Vice versa, evolutionary biologists ignored ecological processes as these were assumed to occur at such short time scales that their effects would be unnoticeable at the long, evolutionary timescales. However, over the past decades, notions have changed from “nothing in biology makes sense except in the light of evolution” [1], to “nothing in evolutionary biology makes sense except in the light of ecology” [2], to finally “nothing in evolution or ecology makes sense except in the light of the other” [3].
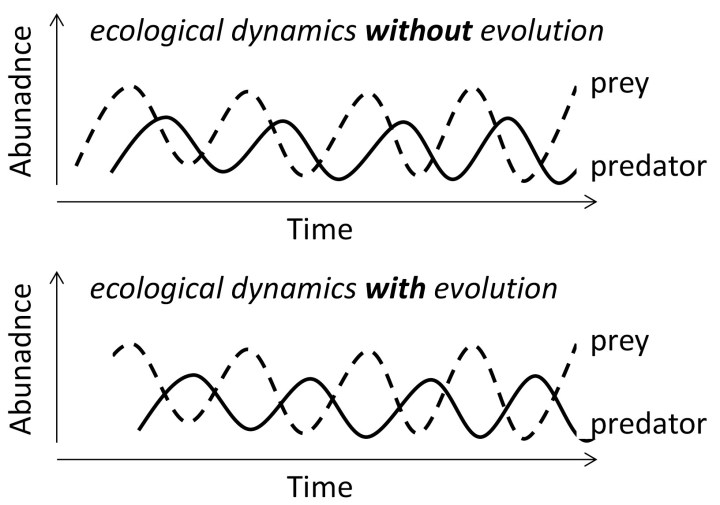
Most studies on eco-evolutionary dynamics have focused on interactions between predators and their prey, like Yoshida and coworkers [4], or between parasites and their hosts. Surprisingly little is known about how eco-evolutionary interactions affect trait dynamics within single populations. Within single populations, it is likely that ecological and evolutionary variables are both the drivers and the objects of change. This means that we can distinguish an ecology-to-evolution pathway and an evolution-to-ecology pathway that together comprise an eco-evolutionary feedback loop (Figure 2).
Discrete alternative phenotypes that occur within single populations present ideal test beds to investigate how eco-evolutionary feedback loops operate. The reason for this is that the alternative phenotypes typically exhibit contrasting growth, reproduction or survival rates so that they are differentially affected by selection (the ecology-to-evolution pathway); in turn, an evolutionary shift in alternative phenotype expression would elicit an ecological response precisely because these alternative phenotypes differ in their demographic rates (the evolution-to-ecology pathway).

Alternative reproductive tactics (ARTs) are an extreme form of discrete phenotype expression within single populations, and occur across many taxa. An excellent example are the male fighters and scramblers of my favourite study species, the bulb mite (Rhizoglyphus robini). Fighter bulb mites possess weapons for male-male competition over access to mates, whereas scramblers are defenceless. Fighters can also mortally injure conspecifics. This is also what my then Master student Jasper Croll found in 2015 in an experiment where he had observed that fighters are more likely to kill juveniles predisposed to develop into fighters [5]. Jasper next took on the challenge of using a population model to investigate if fighter expression can feed back to affect population size and structure, thereby altering the evolutionary dynamics of ART expression in an eco-evolutionary feedback loop.
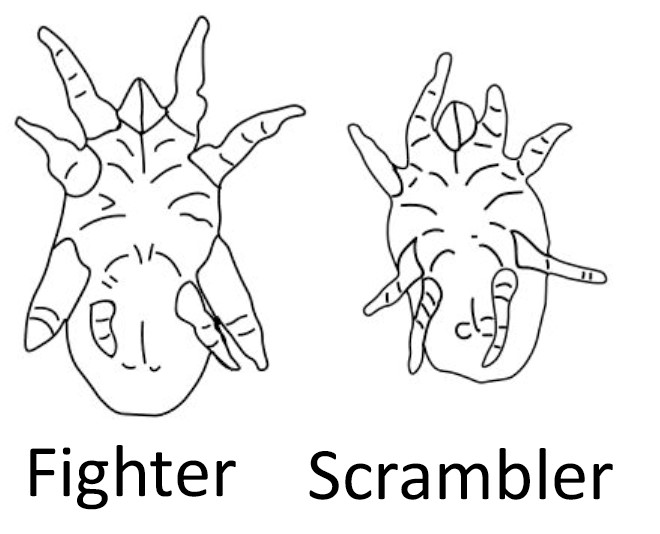
Jasper found that the intraspecific killing by fighters can extend the conditions under which ARTs evolve because fighters that kill other fighters decreases fighter fitness. Importantly: this effect can be nullified when benefits from killing are incorporated, like increased reproduction through increased energy uptake. These results from Jasper’s work is now published in the Journal of Animal Ecology [6], and form part of a Special Feature entitled “The diversity of eco-evolutionary dynamics: comparing the feedbacks between ecology and evolution across scales”; a cross-journal special feature from the British Ecological Society, edited by Franziska Brunner, Jacques Deere, Martijn Egas, Christophe Eizaguirre and Joost Raeymaekers.
Are there still aspects of eco-evolutionary feedback loops that we do not understand? Yes! In my view, the next big challenge is to tease apart both pathways within an eco-evolutionary feedback loop. Key factors that are required to do so are (i) a solid understanding of how ecology (e.g. population density) affects trait distributions, (ii) a solid understanding of the developmental mechanism underlying trait expression, and (iii) a solid understanding of how evolutionary change affects ecological variables (e.g. population density).
And how can we use all this knowledge? It should lead to a solid understanding of how an environmental perturbation, such as a change in precipitation, temperature, predation pressure, etc., affects the two pathways of an eco-evolutionary loop: does a perturbation mainly affect ecological variables, with knock-on evolutionary consequences through the ecology-to-evolution pathway, or would it mainly result in an evolutionary shift in trait expression? Or perhaps both are affected simultaneously? These are essential insights to understand the dynamics of the natural world around us, and how they respond to the ever greater changes in the environment.
References
1 Dobzhansky, T. 1973. Nothing in biology makes sense except in the light of evolution. Am. Biol. Teach. 35: 125-129.
2 Grant P, Grant R. 2008. How and Why Species Multiply: The Radiation of Darwin’s Finches. Princeton Univ. Press, Princeton, NJ.
3 Pelletier F, Garant D, Hendry AP. 2009. Eco-evolutionary dynamics. Philos. Trans. R. Soc. B 364: 1483-1489.
4 Yoshida T, Jones LE, Ellner SP, Fussmann GF, Hairston NG Jr. 2003. Rapid evolution drives ecological dynamics in a predator-prey system. Nature 424: 303-306.
5 Smallegange IM, Fernandes RE, Croll JC. 2018. Population consequences of individual heterogeneity in life histories: overcompensation in response to harvesting of alternative reproductive tactics. Oikos 127: 738-749.
6 Croll JC, Egas M, Smallegange IM. Online. An eco-evolutionary feedback loop between population dynamics and fighter expression affects the evolution of alternative reproductive tactics. Journal of Animal Ecology. DOI 10.1111/1365-2656.12899
*A similar version of this post was posted on isabelsmallegange.com.
